Introduction
Nanoparticles (NPs) are now principal constituents in the biomedical area, especially in drug delivery, imaging, and therapy. NPs are materials with diameters less than 100 nm, and they display outstanding physical and chemical characteristics that vary from the bulk form, with their key feature ascribed mainly to their high surface area-to-volume ratio (1, 2). This special feature allows increased interaction with biological systems, and therefore, NPs can be employed in various biomedical applications, including drug targeting and imaging diagnostics (3, 4). The forms of NPs are diverse, from carbon-based NPs to metal NPs, ceramic NPs, semiconductor NPs, polymeric NPs, and lipid-derived NPs (5). Every type has specific characteristics that are used in particular applications. For example, metal NPs like gold and silver are commonly used for their optical properties in imaging applications, whereas polymeric NPs are used as carriers for hydrophobic pharmaceuticals (6, 7). NPs, though promising, tend to be faced with issues of stability and bioavailability in biological systems. One of the shared issues is Ostwald ripening, which involves the agglomeration of smaller NPs to create larger, less stable particles, resulting in a reduction in functionality (8, 9). Encapsulation of NPs tends to be a possible solution to overcome the mentioned constraints (10, 11). Virus-like particles (VLPs), with identical virus-like morphology without genetic material, provide a protective environment for NPs, with greater stability and bioavailability and also delivery potential to the target site (11). Encapsidation takes advantage of viral capsid protein (CP) self-assembly and creates stable nanocarriers for which there can be specific therapeutic tailoring. For example, cowpea mosaic virus (CPMV) VLPs have been shown to selectively target tissues, and tumors via mechanisms such as the enhanced permeability and retention (EPR) effect (12). This built-in targeting capability, coupled with the capability to encapsulate various functionalized NPs, renders VLPs promising drug delivery and imaging systems. Furthermore, the addition of functionalized NPs, i.e., quantum dots and iron oxide NPs, in VLPs has introduced novel avenues for theragnostic applications, wherein therapeutic drugs and imaging contrast agents are delivered jointly (13). The ability of VLPs in response to various environmental conditions, i.e., pH, also extends the horizon of VLPs in precision medicine (14).
Compared to other major systems like liposomes and dendrimers commonly used as vehicles, VLPs are highly structural and stable, making it highly desirable nanocarrier systems. On the contrary nanocarriers like liposomes, though clinically successful, lack structural stability and can go through rapid clearance without PEGylation, a process to reduce cross-contamination and dendrimers, other widely used NPs, even with the high drug-loading potential, lacks safety in its complex synthesis process, making VLPs a more desired option. Another major advantage of VLPs over the other NPs is that they provide natural biocompatibility, enzyme resistance, and multivalent display capacity, making them highly preferred for both targeted therapy and vaccine preparation, and its ability to encapsulate inorganic, hydrophobic, or sensitive payloads with precision make them highly recommended for next-generation carriers for therapeutic application. Table 1 explains the advantages of VLPs compared to dendrimers and liposomes.
Therefore, the encapsidation of NPs in VLPs is a step forward for nanomedicine, with increased stability, targeted delivery, and multifunctionality. As development focuses on the synthesis and use of VLP-encapsulated NPs, prospects for novel therapeutic treatments with better patient outcomes in diverse medical disciplines become more promising. This review focuses on the build of VLPs, various methods of synthesis, and their mechanism of action. The aim of the review is to bring out a comprehensive study of VLPs, which will help in enhancing futuristic medicine.
Synthesis CP
VLPs are non-infectious particles made up of CPs that self-assemble into virus-like structures that imitate the native virion, but without the viral genome. These VLPs consist only of CPs, which may be synthesized through different expression systems, some of which include microorganisms like Escherichia coli and Baculovirus. The monomers of the capsid obtained from these organisms have the capability for self-assembly. To overcome the limitations of the conventional expression systems, a eukaryotic Pichia pastoris system is frequently used for viral structural protein expression. This yeast system enables the production of high levels of soluble viral structural proteins in large quantities by fermentation. Viral structural protein genes are expressed, and products thus derived are purified and characterized. VLPs from this system have the same structure as that of the native wild-type Cowpea Chlorotic Mottle Virus (CCMV) VLPs (15). The process of their synthesis is provided below (16, 17).
CP expression: The CP coding sequences of the target virus (e.g., CCMV) were cloned into a suitable expression vector and transfected into Pichia pastoris cells. The cells were cultured in a suitable medium, usually 30°C, until they reached an optical density (OD600) of about 0.6.
Induction of Protein Expression: Addition of methanol to the culture medium induces expression of the CPs as a carbon source and the best production of proteins. The incubation is prolonged for 24–48 h to enable maximum protein production.
Harvesting and Lysis: After induction, cells are harvested via centrifugation at 4,000 rpm for 20 min at 4°C. The pellet of cells is subsequently suspended in lysis buffer (50 mM Tris-HCl, pH 8.0, 150 mM NaCl, 1 mM EDTA) and lysed by sonication or enzymatic treatment.
Purification of CPs The lysate was clarified by centrifugation at 12,000 rpm for 30 min, precipitation with ammonium sulfate (30 ± 60% saturation), and dialyzed against buffer to remove salts. The protein was further purified by size-exclusion chromatography (SEC) using a Superdex 200 column.
Characterization of CPs: Purified CPs can be characterized using different techniques such as Sodium Do decyl Sulphate PolyAcrylamide Gel Electrophoresis (SDS-PAGE) to determine protein presence and purity and dynamic light scattering (DLS) to determine the size distribution and polydispersity index (PDI) of CPs.
CP self-assembly into VLPs under the best conditions is normally in a buffer at 20 mM Tris-HCl (pH 7.5) and 100 mM NaCl. The process of assembly may be followed by transmission electron microscopy (TEM) to ensure the morphology and size of the VLPs produced (18). The homogeneous spherical VLPs developed using E. coli and P. pastoris were confirmed using TEM, as shown in Figure 1. The T-number that defines the structure of CP subunits in the capsids of icosahedral viruses is provided by the equation (1) (19):
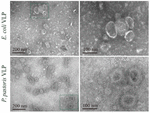
Figure 1. Transmission Electron Microscopy (TEM) image of polymers, E. coli VLPs, and P. pastoris VLPs (18).
where D is the diameter of the viral capsid shell and d is the distance between two adjacent capsid subunits (measured from their centers) (20). This T-number denotes the CP arrangement and has a double function: it stabilizes the material inside the shell as a container and gives a surface to modify the structure of the CP to target specific tissues (2, 19). Depending upon the T-number, the number of NPs that can be encapsidated within the VLPs changes. Figure 2a displayed NPs encapsidated in VLPs with T = 3, having an average of 3 NPs per VLP. However, with T = 1 symmetry, the average no. of NPs encapsidated within VLPs changes to 1.5 (Figure 2b) (19).
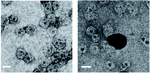
Figure 2. TEM images of encapsidated nanoparticles (NPs) in VLPs with T = 3 and T = 1 (19).
VLP assembly
VLPs have attracted much attention in recent years because of their promising applications in biomedical fields, such as vaccine development and drug delivery. The earliest observation of VLPs was in 1968, when their occurrence was observed in the serum of patients suffering from Down syndrome, leukemia, and hepatitis (11, 22). The various types of capsid structure and its assembly are summarized in Table 2.
Principle
The VLPs can be self-assembled using CPs to produce empty shells with the absence of core genetic material (11, 22). Self-assembly of VLPs is a process based on the intrinsic properties of viral CPs. These proteins have an inherent capability of interacting among themselves for capsid structure formation. The assembly may take place spontaneously under suitable conditions. One of the central mechanisms of VLP assembly is the function of nucleic acids. In most viruses, the presence of the viral genome or other negative charges such as ribo nucleic acid (RNA) or de oxy ribo nucleic acid (DNA) plays an important role in capsid formation. These molecules can serve as a scaffold, where CPs are assembled around them. VLPs, however, may also assemble without nucleic acids, producing empty capsids. The level of CPs is frequently required to be increased in such instances in order to compel the assembly process (25).
Methods of assembly
A number of methods have been established to manage and maximize VLP assembly. These methods can generally be defined as in vivo and in vitro methods (16, 26).
In Vivo Assembly: This method entails expressing the viral CPs in a host organism (16). The CPs are produced and self-assembled as VLPs inside the host cell. If the VLPs are produced in vivo, however, the danger of encapsidating cellular components like host cell proteins is presented and may interfere with the biological function of the VLP (27).
In vitro production of VLPs is a process in which host organisms’ CPs are synthesized and purified but not assembled inside of the host organism. They are harvested as non-assembled CPs to prevent contamination with host-cell products (16, 27). After they are generated, they are put in certain chemical conditions to self-assemble into a capsid. An advantage of in vitro assembly is that different viruses’ CPs can be together and make a hybrid VLP (16).
Control of encapsidation strategies
One area of strong emphasis in VLP studies is the controlled encapsidation of molecules inside the VLP capsid. Various strategies have been designed to do so. They are as follows:
Oligonucleotide-Mediated Assembly: One of the methods is the utilization of a particular oligonucleotide sequence, which binds to the CP as well as the content molecule (21). For instance, red clover necrotic mosaic virus (RCNMV) assembly can be artificially triggered through the synthesis of a DNA oligonucleotide that is identical to RNA2 and hybridizing it with RNA1 to create an Overlapping Assembly Sequence (OAS) (21). This OAS is the same as the native OAS utilized by the native virus during CP assembly. It may therefore spontaneously trigger capsid assembly by anchoring the DNA oligonucleotide onto an NP of preference (21, 28).
Electrostatic Interactions: A different approach uses electrostatic interactions between the content and the CP. Since the inside surface of the viral capsid is positive, by applying a content that is attached to a negatively charged nucleic acid, it provokes the protein assembly (29–31).
Genetic Fusion: A strong approach is genetically attaching the peptide sequence to the NPs of interest that facilitates capsid assembly. It is a superior method since it can be amplified for a wide variety of proteins. It can also be applied for inorganic NPs as carriers for different imaging and diagnostic methodologies (31, 32).
Factors affecting VLPs assembly
There are various factors that affect the assembly of VLPs; they are pH, ionic strength, temperature, and CP concentration. A minimum concentration of CPs is typically required for efficient capsid formation (16).
Special encapsidation techniques and methods
Encapsidation of functionalized NPs
This approach generally employs functionalized gold NPs, e.g., citrate- or tetraethylene glycol (TEG)-coated, to promote encapsidation within viral capsids such as CCMV. Functionalizing NPs to present negative charges, e.g., using carboxylate-terminated polyethylene glycol (PEG) chains. The artificially coated negative NPs provide the optimal environment to induce capsid assembly. For example, brome mosaic virus (BMV) can be functionalized with carboxylate-terminated PEG chains, which impart a negative charge to the NPs to mimic the nucleic acid (33–35). This allows capsid assembly. The BMV capsid can be produced in different sizes and shapes based on the size and shape of the core-encapsidated NPs. Smaller NPs lead to smaller BMV capsid shells. It demonstrates that while VLPs are claimed to be rigid, they can also be flexible in terms of size (36). The yield of encapsidation depends upon the charge, i.e., the number of fully encapsidated viral shells over empty shells. The distinctive feature is the improvement in efficiency of encapidation and decrease in the occurrence of empty shells, with greater control over NP loading (34). The technique also facilitates the encapsulation of a broad range of inorganic NPs using scalable peptide conjugation methods (31). The limitation of this technique is the functionalization might be complicated and expensive, limiting scalability and reproducibility on various types of NPs (31).
Virus shells as nanoreactors
The nanoreactor approach employs preassembled viral capsid shells as confined bioreactors, where the loaded molecules, even metal NPs, are synthesized in situ. CCMV shells, for instance, can be selectively opened to take up negatively charged paratungstate ions when in “open” form, favoring crystallization and growth inside the capsid interior (37). Reversible gate-controlled pore gating provides excellent control over content molecule entrance and confinement, hence preventing undesirable macromolecule contamination (20), offering a perfect microenvironment for controlled synthesis and growth of NPs, and utilizing the selective permeability of the capsid to manage molecule entry (37). Icosahedral viral shells have been used to trap NPs, which were then allowed to nucleate. In this case, the NPs trapped within could be used for imaging and detection purposes while the biocompatibility of the VLP was provided by the viral capsid shell (35). The significance of this method is that it also supports direct synthesis of NPs with specified size and morphology restricted by the interior of the capsid (20). The drawback of this technique is that it is limited to the loaded molecules and reagents that can access and react in the confined capsid interior, making it less generally applicable (12).
Gating of VLPs
The gating process exploits the pH-dependent structural behavior of VLPs, in which capsid shells swell at elevated pH, expanding pore size and accommodating small negatively charged molecules or NPs to penetrate into the lumen (38). With the reduction of pH, the capsid shrinks, encapsulating molecules inside. For instance, negatively charged TiO2 NPs can penetrate CCMV shells via this gating process. The benefit is that it enables temporally controlled encapsidation and cargo release depending on environmental pH, and this can be fine-tuned specifically for targeted delivery systems. The drawback of this technique is their dependence on specific pH conditions limits its application in physiological conditions where pH variation is limited or tightly controlled (38). Table 3 compares the differences between different types of encapsidation techniques to encapsidate NPs.
Validation of encapsidation of NPs
For assessing the efficiency of encapsidation of NPs in VLP, many techniques are used. Previously conventional imaging techniques like TEM, DLS, and many analytical strategies were used to monitor and quantify the process. Even instruments like ultra violet (UV) spectroscopy are characterized to detect shifts in plasmon resonance during encapsulation in the presence of NPs. Further separation techniques like electrophoresis and density gradient ultracentrifugation are also applied to calculate the integrity and success of the encapsidation process. All the above-mentioned methodologies together contribute to the standard controls of VLPs in a border spectrum.
Mechanism of action of encapsulated NPs
The mechanism of the encapsidated NPs to attain their biological activity is mainly mediated through their cellular internalization and VLP-mediated protection. VLPs composed of viral CPs create a protective protein layer over NPs to safeguard them from degradation or aggregation occurring in the early stages, enhancing their stability and bioavailability (11, 35, 39).
Cellular uptake pathways
Encapsidated NPs enter cells primarily through receptor-mediated endocytosis, in which VLPs bind to cell surface receptors and are taken into vesicles known as endosomes. These vesicles fuse with lysosomes, where degradation is expected to take place. But VLP-encapsidated NPs have also been shown to evade or escape lysosomal degradation, allowing more efficient delivery of therapeutic or diagnostic payloads into the cytoplasm (11, 39, 40). Besides that, some VLPs can enter cells by non-endocytic pathways, such as direct fusion with the cell membrane, broadening their delivery range (39). This two-way entry makes intracellular transfer and release of the material they hold effective.
Protection and stability
VLP encapsulation protects NPs from biological and chemical degradation like enzymatic degradation and aggregation. For example, quantum dots encapsulated in VLPs are retained in fluorescence and are resistant to aggregation, and gold NPs stabilize and exhibit higher functionalization potential (22, 35). Figure 3 exhibits the encapsidation of Au NPs by CCMV CP, where the spherical shape of the Au NPs induces the CP to assemble into a spherical shape around the NPs. Figure 4 displayed that VLPs can contain 1–10 nm particles, and also, more than one NP can be encapsidated in a single VLP. This protective action ensures the functionality of NPs in intricate biological environments.

Figure 3. TEM images of 18 nm Au NPs encapsidated by the Cowpea Chlorotic Mottle Virus (CCMV) VLPs at different pH (22).

Figure 4. TEM images of 5 nm Au NPs (1 to many NPs) encapsidated by the CCMV VLPs (22).
Targeting and specificity
The viral capsid origin offers an innate targeting ability to VLPs, since capsids still exhibit tropism to particular tissue/cell types. For example, CPMV-based VLPs selectively accumulate in the liver, kidney, and spleen, whereas Hepatitis B virus (HBV)-based VLPs specifically target liver cells (11, 12). This intrinsic targeting can be further supported by bioconjugation of ligands such as transferrin or cell-specific peptides to the capsid surface, which improves selectivity and uptake by diseased tissues, including EPR tumors (11, 13).
Controlled release and environmental responsiveness
Some of the VLPs present conformational variations based on external cues, e.g., pH. For example, CCMV illustrates gate mechanisms for opening the pores of the capsid at elevated pH, allowing for controlled diffusion of negatively charged payload molecules like TiO2 NPs inside, followed by entrapment when the pH is reduced (38). This feature offers a mechanism for controlled encapsidation and release of drugs dependent on intracellular or extracellular conditions.
Discussion
Different NPs can be enclosed within VLPs of diverse viral origin to produce encapsulated NPs with enhanced drug delivery and imaging applications. These encapsidated NPs have been proven to show higher efficiency, biocompatibility, and stability when compared to traditional drug delivery (22). When administered to an individual, CPMV VLPs were found to be concentrated in the liver, kidney, spleen, and blood (41). They were detected by fluorescence spectroscopy and real time polymerase chain reaction (RT-PCR). The viral particles were found to be intact, showing their resistance against low gastric pH and digestive enzymes (42, 43). Owing to its characteristics, CPMV is a good biocompatible candidate for bioimaging, such as computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET), and optical modalities. It is used to produce highly sensitive images of vasculature in a technique known as intravital imaging (12, 41). NPs produced through the conventional methods are highly unstable (nanoshells) and cytotoxic (nanorods). When Au NPs are encapsulated using CCMV shells, they demonstrate increased stability and can be easily amplified, assembled, and disassembled (22). NPs such as quantum dots can be encapsidated within VLPs to prevent aggregation and can be used in imaging techniques. This is because each NP is compartmentalized with its own viral shell and hence cannot aggregate with other particles. It also produces VLPs that have the scope to be bioconjugated with ligands that help to target specific tissues (35). Furthermore, theranostics is a concept that uses these viral shells to encapsulate both therapeutic and imaging molecules (therapeutics and diagnostics). For example, VP4 rotavirus VLPs that are loaded with doxorubicin and iron oxide NPs simultaneously enable them with dual function: they help in imaging and selective killing of cancer cells (44). VLPs are advantageous as they are able to deliver NPs to a particular site due to the innate property of a virus in targeting a tissue; that is, the origin of the viral capsid determines its specific affinity for a target cell. For example, the HBV infects liver cells usually, whereas HBV-derived VLPs specifically target liver cells. Hence, they are ideal for targeted delivery of drugs and diagnostic molecules (11). As viral shells are made of viral proteins, they can induce an immune response (e.g., adenovirus), which is commonly utilized in gene therapy and has been linked to severe hepatotoxicity. Although these viral shells are not derived from human pathogens nor do they replicate in vivo, it is necessary to screen for potential immune responses. Studies have shown that assembled virus shells are more immunogenic than the monomeric units of capsid.
VLPs are structurally and visually alike to their host live viruses but lack the complete genome of the virus and are non-infectious. This structural variation adopted by different VLPs makes them structurally and functionally attractive in vaccine delivery. Based on the presence or absence of lipid envelopes, VLPs are classified mainly into two types: enveloped and non-enveloped, and further divided as single-layer and multilayer based on the organization of CPs, which inherit the immune responses. Previous studies have highlighted the VLPs, due to their immunogenic effect, might lead to antibody clearance, which was controlled by PEGylation and surface modification with immuno-inert polymers that can reduce immunogenicity. Moreover, plant- or bacteriophage-based VLPs, which are commonly used, are biologically safe and self-adjuvanting, making it desirable in vaccine preparation. They possess no risk of human pathogen homologues, and robust immune responses are noted in these cases, but immunotoxicological assessments and cytokine profiling should be included with in vivo studies to monitor the compatibility with human administration.
Studies have also shown that PEG-coated virus shells have lesser immunogenicity, as they reduce the antibody production against these virus particles (12). Metal NPs, magnetic NPs, and fluorescent quantum dots (semiconductor NPs) are the most commonly used NPs in imaging applications (35). Magnetic NPs are used in various biomedical fields such as MRI, cell sorting, and probing using bio-probes. Here, the magnetic NPs are designed to be superparamagnetic, which refers to NPs with reversible magnetic fields. The superparamagnetic NPs, such as iron oxide NPs, are negatively charged and are encapsidated within the positively charged BMV shell. Furthermore, iron oxide NPs can be coated with carboxy-terminated PEGylated phospholipids, which impart both water solubility and various functionalities for biomedical applications. This leads to the formation of micelle-coated NPs, which are easily encapsulated by CPs to form BMV with magnetic cores (45). CCMV shells undergo gating mechanisms and can entrap magnetic iron oxide NPs, which can be used as an MRI contrasting agent, while those with fluorescent cores can be used for fluorescent spectroscopy (35). This property of CCMV can be attributed to the presence of metal binding sites in the interior surface of the CP. Apart from CCMV M13 phage, simian virus 40 and BMV were also discovered to have the ability to encapsidate iron oxide NPs for use as contrasting imaging agents (44). The metal binding site has the ability to attach to lanthanides such as terbium (III) and gadolinium apart from iron oxide NPs (12). Terbium (III) and gadolinium can also be sequestered within RNA-containing capsids of CPMV. The nucleic acid is required in order to attach the ions to its binding site. As the affinity between gadolinium and the coat proteins is weak, it is not used for in vivo imaging techniques, as these ions can enter body tissue and cause toxicity. But similar encapsulated lanthanide complexes can be used as effective MRI reagents (35). P22 virus was used as the template for the synthesis of TiO2 and iron oxide NPs by incorporating an anionic peptide sequence to the interior CP. This induces the synthesis of the respective metal NPs, and the particle continues to grow within the VLP until there is no more space left. Similarly, HBV-based VLP was modified by attaching histidine residues to the N-terminal, which resulted in increased binding of iron oxide NPs to the interior surface of the capsid shell. These HepB molecules containing a magnetic core were discovered to be more readily taken up by cells (46). VLPs can also be used as templates for the synthesis of nanowires because they provide a high degree of uniformity, stability, and chemical functionality. These viruses can be genetically modified to include specific peptide sequences that can easily bind and interact with the cargo molecules. Nanowires are synthesized within VLPs derived from helical viruses. As M13 phage had the ability to alter functional capsids for newer and better applications, it was used as the template. M13 phage was used due to its easy production and genetic tractability. The P8 phage proteins were modified to help improve binding with gold particles. The P8 proteins, which are present along the length of the M13 phage, thus help to template the synthesis of gold and gold-platinum nanowires. By inducing mutations on the capsid genome, a variety of CPs can be synthesized for the desired effect, e.g., mutations in the TMV genome allowed for longer capsid monomers, which assembled with an identical diameter to that of the native phage. This modified phage is further altered by reversing the negatively charged interior surface and binding different cysteine sequences to the N-terminal, allowing for the production of palladium NPs (44). Encapsulated NPs can undergo extravasation easily in tumor vasculature, as they have leaky or increased fenestrations, and eventually accumulate at the tumor site by the process of EPR. As the tumor tissue and the tumor vasculature are morphologically different than the normal tissue, they are easy targets for antineoplastic treatment and diagnostics. CPMV particles were found to accumulate at the tumor site after 24 h of administration in mouse models with tumor xenografts. This selectivity in accumulation is due to EPR and can be enhanced by binding certain ligands to the VLP surface (13). CPMV particles used for targeting tumor cells were conjugated with transferrin, which is a protein required for iron uptake. The conjugated VLP binds to the transferrin receptors, which are usually up-regulated in tumor cells, as iron is required in excess in these tissues. On the contrary, M13 phage particles are used to target tumor vasculature. These phage particles were conjugated with chemotherapeutic drugs and were shown to target the vasculature (12).
Besides such advantages, there are still some challenges in the development and use of VLP-encapsidated NPs. One of the main challenges is the immunogenicity of viral proteins. Although VLPs are non-infective, they have the ability to induce immune responses, which may limit their potential clinical applications. It has been shown through studies that PEGylation of VLPs may decrease their immunogenicity by preventing excessive antibody production against these viral particles (12). Therefore, optimizing the surface characteristics of VLPs to enhance both targeting efficiency and immunogenicity to the maximum for effective use in the clinic is pertinent. Large-scale production of VLPs and encapsulation of NPs is another challenge. Although in vitro assembly methods offer controlled environments and reduced contamination, they are likely to require special conditions that are hard to replicate on a large scale (16). Conversely, in vivo assembly methods, though yielding high titers of VLPs, are prone to host cell product contamination, which can affect their biological activity (27). Stable and scalable production processes with high encapsidation efficiency but low empty shell content should be developed to move this technology forward. Comprehensive toxicity research of the encapsulated NPs should be conducted to verify their safety for clinical application. Despite the fact that VLPs form a protective sheath for NPs, long-term in vivo implications of these systems are still yet to be realized. Research in the future should focus on extensive studies of toxicity and designing hybrid VLP systems that utilize synergistic effects of different viral proteins and NPs to develop more efficient and multivalent drug delivery systems.
In nanomedicine, the features of NPs are a major concern; the therapeutic efficacy of it solely depends on the interaction and fate in biological systems and their safety. Features such as the size, shape, and biochemical properties of NPs can vary based on methodology (i.e., synthesis method), environmental (i.e., storage time), and commercial (i.e., laboratory supplier) factors. Till date, 110 viral proteins from 35 viral families have been shown to be capable of assembly into the VLPs. Several VLP-based vaccines for human use, including Recombivax HB and Engerix-B for HBV, Gardasil, Cervarix, and Gardasil-9 for HPV, and Hecolin for HEV, have been licensed for clinical use.
VLP-based preparations are rigorously characterized by several preclinical methods commonly used in inorganic nanotechnology and protein chemistry. For accurate clinical use, VLP preparation must incorporate good laboratory practice, compliant toxicology, pharmacokinetic modeling, and clinical-grade manufacturing protocols. Analytical protocols should be developed for nanosized materials and protein chemistry for VLP characterization, which will help in fulfilling precision medicine goals and justify the use of VLPs as nanocarriers for therapeutic applications.
Conclusion
Encapsulation of NPs within viral capsid shells has many advantages, such as higher stability, targeted delivery, and protection of the components against degradation. Key results show that all the different encapsidation techniques, including viral capsid assembly, in vitro and in vivo assembly, and functionalized NP encapsulation, have their own advantages and disadvantages that need to be weighed cautiously upon choosing an optimal approach for particular biomedical applications. Even though the field has been promising with recent developments, there are various challenges that persist. Future studies should be directed at enhancing encapsidation efficiency to maximize drug loading capacity with minimal empty shell presence. Moreover, whether encapsulated NPs are toxic is a concern to address to ensure safety in clinical use. The investigation of new hybrid VLP systems in terms of synergy benefits of different viral proteins and NPs can lead to the development of more efficient and multifunctional drug delivery systems. From these studies, we are able to further exploit the potential of encapsulated NPs, bringing with it new therapeutic possibilities with great potential for significantly improving patient care in various fields of medicine.
VLPs are one of the promising encapsidation methods, but there are some technical challenges, such as changes in conditions such as temperature, shear force, and process that are used in purification that can destroy the integrity and stability of VLPs alongside reducing its immunogenicity. Moreover, high production costs, particularly in protein purification and expression, remain a barrier to large-scale VLPs production issue, and in some situations further treatments may be required to reduce the levels of unwanted contaminants, which might, in long run, risk the antigenicity of VLPs itself. All these limitations are to be considered in producing VLPs. Thus, major challenges for future studies on VLPs should assess their safety, stability, integrity, potential toxicity, and immunogenicity against variable temperatures and pHs, which can be overcome through advanced bioengineering, automated assembly, and standardized quality control measures.
References
1. Khan, Y, Sadia, H, Ali Shah, SZ, Khan, MN, Shah, AA, Ullah, N, et al. Classification, synthetic, and characterization approaches to nanoparticles, and their applications in various fields of nanotechnology: a review. Catalysts. (2022) 12:1386.
2. Tiwari, G, Tiwari, R, Sriwastawa, B, Bhati, L, Pandey, S, Pandey, P, et al. Drug delivery systems: an updated review. Int J Pharm Investig. (2012) 2:2–11. doi: 10.4103/2230-973X.96920
3. Yusuf, A, Almotairy, ARZ, Henidi, H, Alshehri, OY, and Aldughaim, MS. Nanoparticles as drug delivery systems: a review of the implication of nanoparticles’ physicochemical properties on responses in biological systems. Polymers (Basel). (2023) 15:1596.
4. Jeevanandam, J, Barhoum, A, Chan, YS, Dufresne, A, and Danquah, MK. Review on nanoparticles and nanostructured materials: history, sources, toxicity and regulations. Beilstein J. Nanotechnol. (2018) 9:1050–74. doi: 10.3762/bjnano.9.98
5. Khan, I, Saeed, K, and Khan, I. Nanoparticles: properties, applications and toxicities. Arab J Chem. (2019) 12:908–31. doi: 10.1016/j.arabjc.2017.05.011
6. Burlec, AF, Corciova, A, Boev, M, Batir-Marin, D, Mircea, C, Cioanca, O, et al. Current overview of metal nanoparticles’ synthesis, characterization, and biomedical applications, with a focus on silver and gold nanoparticles. Pharmaceuticals. (2023) 16:1410.
7. Khodabux, R, Parvathi, V, and Harikrishnan, T. Nanocurcumin: potential natural alkaloid against oral squamous cell carcinoma. Biomed Biotechnol Res J. (2021) 5:252–9. doi: 10.4103/bbrj.bbrj_102_21
8. Gawande, MB, Goswami, A, Felpin, F-X, Asefa, T, Huang, X, Silva, R, et al. Cu and Cu-based nanoparticles: synthesis and applications in catalysis. Chem Rev. (2016) 116:3722–811.
9. Gommes, CJ. Ostwald ripening of confined nanoparticles: chemomechanical coupling in nanopores. Nanoscale. (2019) 11:7386–93. doi: 10.1039/C9NR01349K
10. Shahrivarkevishahi, A, Hagge, LM, Brohlin, OR, Kumari, S, Ehrman, R, Benjamin, C, et al. Virus-like particles: a self-assembled toolbox for cancer therapy. Mater Today Chem. (2022) 24:100808.
11. Nooraei, S, Bahrulolum, H, Hoseini, ZS, Katalani, C, Hajizade, A, Easton, AJ, et al. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. J Nanobiotechnol. (2021) 19:59. doi: 10.1186/s12951-021-00806-7
12. Manchester, M, and Singh, P. Virus-based nanoparticles (VNPs): platform technologies for diagnostic imaging. Adv Drug Deliv Rev. (2006) 58:1505–22. doi: 10.1016/j.addr.2006.09.014
13. Rohovie, MJ, Nagasawa, M, and Swartz, JR. Virus-like particles: next-generation nanoparticles for targeted therapeutic delivery. Bioeng Transl Med. (2017) 2:43–57. doi: 10.1002/btm2.10049
14. Tsvetkova, IB, and Dragnea, BG. Encapsulation of nanoparticles in virus protein shells. Methods Mol Biol. (2015) 1252:1–15. doi: 10.1007/978-1-4939-2131-7_1
15. Sugrue, RJ, Cui, T, Xu, Q, Fu, J, and Cheong Chan, Y. The production of recombinant dengue virus E protein using Escherichia coli and Pichia pastoris. J Virol Methods. (1997) 69:159–69. doi: 10.1016/S0166-0934(97)00151-1
16. Le, DT, and Müller, KM. In vitro assembly of virus-like particles and their applications. Life. (2021) 11:334. doi: 10.3390/life11040334
17. Tomé-Amat, J, Fleischer, L, Parker, SA, Bardliving, CL, and Batt, CA. Secreted production of assembled norovirus virus-like particles from Pichia pastoris. Microb Cell Fact. (2014) 13:1–9.
18. Tian, Q, Huo, X, Liu, Q, Yang, C, Zhang, Y, and Su, J. VP4/VP56/VP35 virus-like particles effectively protect grass carp (Ctenopharyngodon idella) against GCRV-II infection. Vaccines. (2023) 11:1373.
19. Tagit, O, De Ruiter, MV, Brasch, M, Ma, Y, and Cornelissen, J. Quantum dot encapsulation in virus-like particles with tuneable structural properties and low toxicity. RSC Adv. (2017) 7:38110–8.
20. Aniagyei, SE, DuFort, C, Kao, CC, and Dragnea, B. Self-assembly approaches to nanomaterial encapsulation in viral protein cages. J Mater Chem. (2008) 18:3763–74. doi: 10.1039/b805874c
21. Loo, LN, Guenther, RH, Basnayake, VR, Lommel, SA, and Franzen, S. Controlled encapsidation of gold nanoparticles by a viral protein shell. J Am Chem Soc. (2006) 128:4502–3. doi: 10.1021/ja057332u
22. Durán-Meza, AL, Escamilla-Ruiz, MI, Segovia-González, XF, Villagrana-Escareño, MV, Vega-Acosta, JR, and Ruiz-Garcia, J. Encapsidation of different plasmonic gold nanoparticles by the CCMV CP. Molecules. (2020) 25:2628. doi: 10.3390/molecules25112628
23. Bancroft, JB. The self-assembly of spherical plant viruses. Adv Virus Res. (1970) 16:99–134. doi: 10.1016/S0065-3527(08)60022-6
24. Pavithra, AJ, Akshaya, K, Arthi, C, and Saravanan, R. Acquired immunodeficiency syndrome: a walk through from its origin to nanotechnology-based treatment. Arashi J Met Mater Sci. (2024) 1:8–20.
25. Wu, M, Brown, WL, and Stockley, PG. Cell-specific delivery of bacteriophage-encapsidated ricin a chain. Bioconjug Chem. (1995) 6:587–95. doi: 10.1021/bc00035a013
26. Gupta, R, Arora, K, Roy, SS, Joseph, A, Rastogi, R, Arora, NM, et al. Platforms, advances, and technical challenges in virus-like particles-based vaccines. Front Immunol. (2023) 14:1123805.
27. Vicente, T, Mota, JPB, Peixoto, C, Alves, PM, and Carrondo, MJT. Rational design and optimization of downstream processes of virus particles for biopharmaceutical applications: current advances. Biotechnol Adv. (2011) 29:869–78. doi: 10.1016/j.biotechadv.2011.07.004
28. Zahmanova, G, Aljabali, AA, Takova, K, Toneva, V, Tambuwala, MM, Andonov, AP, et al. The plant viruses and molecular farming: how beneficial they might be for human and animal health? Int J Mol Sci. (2023) 24:1533.
29. Strugała, A, Jagielski, J, Kamel, K, Nowaczyk, G, Radom, M, Figlerowicz, M, et al. Virus-like particles produced using the brome mosaic virus recombinant capsid protein expressed in a bacterial system. Int J Mol Sci. (2021) 22:3098.
30. Hagan, MF. A theory for viral capsid assembly around electrostatic cores. J Chem Phys. (2009) 130:114902.
31. Aanei, IL, Glasgow, JE, Capehart, SL, and Francis, MB. Encapsulation of negatively charged cargo in MS2 viral capsids. Methods Mol Biol. (2018) 1773:303–17. doi: 10.1007/978-1-4939-7808-3_21
32. Mardanova, ES, Vasyagin, EA, and Ravin, NV. Virus-like particles produced in plants: a promising platform for recombinant vaccine development. Plants. (2024) 13:3564.
33. Dragnea, B, Chen, C, Kwak, ES, Stein, B, and Kao, CC. Gold nanoparticles as spectroscopic enhancers for in vitro studies on single viruses. J Am Chem Soc. (2003) 125:6374–5. doi: 10.1021/ja0343609
34. Chen, C, Daniel, MC, Quinkert, ZT, De, M, Stein, B, Bowman, VD, et al. Nanoparticle-templated assembly of viral protein cages. Nano Lett (2006) 6:611–5. doi: 10.1021/nl0600878
35. Pokorski, JK, and Steinmetz, NF. The art of engineering viral nanoparticles. Mol Pharm. (2011) 8:29–43. doi: 10.1021/mp100225y
36. Sun, J, DuFort, C, Daniel, MC, Murali, A, Chen, C, Gopinath, K, et al. Core-controlled polymorphism in virus-like particles. Proc Natl Acad Sci U S A. (2007) 104:1354–9. doi: 10.1073/pnas.0610542104
37. Douglas, T, and Young, M. Host-guest encapsulation of materials by assembled virus protein cages. Nature. (1998) 393:152–5. doi: 10.1038/30211
38. Klem, MT, Young, M, and Douglas, T. Biomimetic synthesis of β-TiO2 inside a viral capsid. J Mater Chem. (2008) 18:3821–3. doi: 10.1039/b805778h
39. Dimitrov, DS. Virus entry: molecular mechanisms and biomedical applications. Nat Rev Microbiol. (2004) 2:109–22. doi: 10.1038/nrmicro817
40. Zdanowicz, M, and Chroboczek, J. Virus-like particles as drug delivery vectors. Acta Biochim Pol. (2016) 63:469–73. doi: 10.18388/abp.2016_1275
41. Aljabali, AAA, Alzoubi, L, Hamzat, Y, Alqudah, A, Obeid, MA, Al Zoubi, MS, et al. A potential MRI agent and an anticancer drug encapsulated within CPMV virus-like particles. Comb Chem High Throughput Screen. (2021) 24:1557–71.
42. Shahgolzari, M, Pazhouhandeh, M, Milani, M, Yari Khosroushahi, A, and Fiering, S. Plant viral nanoparticles for packaging and in vivo delivery of bioactive cargos. Wiley Interdiscip Rev Nanomed Nanobiotechnol. (2020) 12:e1629.
43. Rae, CS, Wei Khor, I, Wang, Q, Destito, G, Gonzalez, MJ, Singh, P, et al. Systemic trafficking of plant virus nanoparticles in mice via the oral route. Virology. (2005) 343:224–35. doi: 10.1016/j.virol.2005.08.017
44. Glasgow, J, and Tullman-Ercek, D. Production and applications of engineered viral capsids. Appl Microbiol Biotechnol. (2014) 98:5847–58. doi: 10.1007/s00253-014-5787-3
45. Huang, X, Bronstein, LM, Retrum, J, Dufort, C, Tsvetkova, I, Aniagyei, S, et al. Self-assembled virus-like particles with magnetic cores. Nano Lett (2007) 7:2407–16. doi: 10.1021/nl071083l
46. Shen, L, Zhou, J, Wang, Y, Kang, N, Ke, X, Bi, S, et al. Efficient encapsulation of Fe3O4 nanoparticles into genetically engineered hepatitis B core virus-like particles through a specific interaction for potential bioapplications. Small. (2015) 11:1190–6. doi: 10.1002/smll.201401952
© The Author(s). 2025 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.



