Introduction
Orthopedic implants, such as fracture fixation devices, artificial joints and intervertebral disc prostheses, and bone defect fillers, play a vital role in maintaining, supporting, and restoring the structure and function of the musculoskeletal system. Common implant materials in orthopedics include alloys, ceramics, metals, and polymers, with alloys and metals being the most prevalent (1–3). However, prolonged presence of these implants inside the body encourages infection. Implant-associated infections (IAIs), such as fracture-related infections and prosthetic joint infections, create serious orthopedics complications. Biofilms formed by the microorganisms over implants are very difficult to eradicate owing to the extracellular polymeric substances secreted by the microorganisms, which protects them from the host immune system and antibiotics (4). Combat strategies for IAIs prevention and treatment have been innovated, including antibacterial metal materials development through alloying techniques using materials like cobalt, tantalum, titanium, and biodegradable metals (5, 6). Substantial advancements have been made in creating biodegradable alloys based on iron (Fe), magnesium (Mg), and zinc (Zn) that exhibit antibacterial characteristics, making them suitable for use in orthopedic implants (7).
Bone defects are significant complications often resulting from infections, tumors, or trauma. Recently, tissue engineering has explored alternative scaffold materials, with metals being a key focus. To advance the development of in-vivo testing models that are relevant clinically for evaluating metallic biomaterials in bone defect repair, it becomes crucial to create models that assess their degradation, interactions, and biocompatibility with host tissues. Titanium (Ti) alloys demonstrate exceptional osteocompatibility, biocompatibility, and corrosion resistance, as evidenced from previous research, which makes them promising candidates to be utilized for engineering bone tissue (8). The conventional bone plates are manufactured using titanium alloy or stainless steel, and have shown effective results in treating bone fractures. However, traditional bone plates still face several limitations, such as getting loose and reduced stress shielding, instigated by the modulus difference between the metal implants and tissues of bone, which can hinder optimal healing of the fracture. Furthermore, due to changes in demographic conditions and abnormal loading conditions, the number of patients with complex fractures, like osteoporotic and comminuted fractures, is rising, presenting a significant challenge for the conventional bone plates designed for repairing standard fractures (9).
Currently, biodegradable materials sought significant attention in temporary devices for medical implants, eliminating the need for subsequent implant removal surgery (10). Significant advancements have been made in the research of biodegradable magnesium-based alloys, and coating them with active agents, leading to their increased use in the medical industry (11, 12). These alloys offer several advantages, including reducing reliance on conventional permanent implants made from metals and their biocompatible alloys, such as cobalt-based alloys, stainless steel, and Ti alloys, which typically require another surgery for their removal. These procedures can cause undesirable effects like the release of metal ions and stress shielding. These complications negatively impact patients’ emotional and physical well-being while increasing costs for both patients and healthcare systems. In contrast, biodegradable implants dissolve naturally in the body, eliminating the need for removal after the bone has healed. Magnesium alloys are particularly favored as biodegradable implants in orthopedics because of their natural biodegradation, excellent biocompatibility, low modulus of elasticity (similar to natural bone), and lightweight nature, making them ideal temporary biomaterials (13). Functional gradient materials (FGMs) represent a contemporary class of materials that provide multiple functionalities and can closely replicate the hierarchical and gradient structures seen in natural systems. The human bone structure is anisotropic, meaning it biologically possesses functionally graded properties that vary in different directions. As a result, a variety of orthopedic implants, like knee and hip replacements, as well as bone plates, can perform better if they are FGM. In this regard, the use of additive manufacturing (AM) has greatly advanced the development of FGM for orthopedic applications, enabling the customization of the anisotropic properties (14). Since biomedical implants often replace bone tissue, and our bone itself is an organic, naturally occurring FGM, the application of the FGM concept in implants is quite logical. One of the main benefits of FGMs is their capability to offer customized morphological characteristics, resulting in graded physical as well as mechanical properties along specific directions. These gradual changes in composition, constituents, grain size, microstructure, texture, and porosity stretching towards one or more directions lead to variations in functional properties. This can help address challenges like stress shielding, promote better osseointegration, and enhance both electrochemical performance and wear resistance. Composites of metal and metal-ceramic are a few significant types of metallic FGMs, many of which are specially fabricated for biomedical applications (15). The average expectancy of implant life and the need for reconstruction of bone tissue, both are increasing, warranting the development of load-bearing implantable materials. These materials encourage significant osseointegration and prevent the postoperative infections. To address this challenge, strategies involving surface modification are needed for the metallic load-bearing scaffolds and implants (16). However, while incorporating surface features to improve osteocompatibility can enhance bone integration, it can also raise the risk of infection by promoting the formation of bacterial biofilm. Therefore, the engineering of multifunctional coatings that simultaneously meet these complex and competing demands is critical (17, 18). These coatings must also be versatile, cost-effective, and scalable to facilitate mass production and clinical application (19, 20). In this review we shall discuss about the various functional coatings on biometals and alloys that are developed for being utilized in orthopedic implants. PubMed, Google Scholar, MEDLINE, and the cross-references and references of authors’ lists were searched for mining the related articles published since 2020. The search terms were used primarily were “functional coatings” and “orthopedics.” The search terms that are singly or in combination used included “functional coating” and “orthopedics,” “metals,” “biometals,” “alloys,” “biodegradable materials,” “bioceramics.” Further references were sourced from individual articles.
Process of bone fracture healing
In bone fracture healing, a number of cellular and biomechanical factors interact, including a series of events. Approximately 5–10% of bone fractures do not heal properly, leading to delayed healing or nonunion. The healing of bone fractures can be classified into 2 types: primary healing and secondary healing. The primary healing process involves restoring the bone cortex directly after a fracture, and can only occur if fracture fragments are aligned perfectly and are stabilized internally, with interfragmentary strain kept at minimal levels. The periosteum and surrounding soft tissues participate in secondary healing, resulting in the formation of calluses. Micromotion is beneficial in this type of healing, but stiff fixation can hinder it. A secondary healing process involves both endochondral and intramembranous ossification, and each of these procedures contributes to fracture repair in different ways (21–23).
Hematoma formation occurs in the gap of the fracture within the initial hours after a fracture because of acute inflammation happening in the surrounding soft tissue. Various inflammatory and immune cells populate this hematoma, which releases biological factors that trigger a cascade of cellular events. A small number of osteoblasts and osteoprogenitor cells differentiate into osteoblasts near the fracture site, followed by differentiation into chondrocytes. The hematoma tissue is then gradually replaced by a cartilaginous callus, while intramembranous ossification leads to hard callus formation in the subperiosteal area. As the process progresses, hypertrophicity is seen in the chondrocytes releasing calcium, undergoing apoptosis, and initiating endochondral ossification. The differentiation of monocytes leads the osteoclast-like cells to break down the calcified cartilage, while the mesenchymal stem cells continue towards osteoblastic differentiation, filling the resorption spaces with new bone. This results in the woven bone formation with a trabecular structure, as there is the replacement of the cartilaginous callus with a hard callus. The final stage, bone remodelling, involves coordinated activity between osteoblasts and osteoclasts over several months, during which the fracture callus is remodelled into lamellar bone (24–26). The remodelling of stem cells is known to be directed by the extracellular matrix (27, 28). The bone healing process is shown in Figure 1 [adapted from (29)].
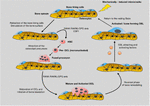
Figure 1. Cycle of bone remodelling processes (29).
Repairing, reconstructing, and replacing congenital malformations, as well as addressing iatrogenic or exogenous tissue and defects in organs, necessitates the use of a wide variety of personalized biomaterials. Mimicking of natural bone healing is executed by the artificial implants, which not only give mechanical support but also accelerate the healing process. Selected metals are known to play a vital role as an implant material (9). In addition to traditional materials like stainless steel, additional options such as pure titanium, titanium alloys, cobalt-chromium alloys, and newer alloy materials, including tantalum-based alloys, are increasingly being utilized in clinical settings. Notably, porous tantalum trabecular metal has gained popularity in the field of orthopedics. Comparisons of surface passivation films across different metals in various environments reveal that tantalum exhibits excellent electrochemical corrosion resistance, with minimal metal ions release and reduced cellular damage. Furthermore, studies in protein adsorption, cytology, molecular biology, and hematology, along with consistent patients’ follow-up observations using porous tantalum trabecular metal, confirm its outstanding biocompatibility. Given its superior biocompatibility and corrosion resistance, tantalum metal holds significant potential for clinical applications (30). Scientists have established that copper (Cu) possesses biological activities that are especially advantageous for the orthopedic biomaterial applications, including implant coatings and biodegradable bone substitutes. Cu has antibacterial properties, promotes angiogenesis, and enhances osteogenesis—key factors for successful biomaterial integration and healing. Copper-doped biomaterials exhibit antibacterial effects, becoming a promising alternative for prophylactic antibiotics and reducing the antibiotic resistance. Additionally, by stimulating the growth of blood vessels and promoting the formation of new bone, Cu significantly enhances the bio-integration of biomaterials, making it an excellent doping agent for orthopedic implants (31). As the field of implants has progressed, the surface functionalization or addition of effective coating has been added to them for improved and effective implants free from bacterial contamination in-vivo.
Interaction between bacteria and orthopedic implant
In IAI, there are 3 major contributors involving bacteria that make the implant unsuccessful.
Surface adhesion of bacteria
Bacteria tend to adhere to material surfaces more readily than to their surrounding aqueous environments. The initial stage is bacterial adhesion on implant surfaces involving chemical and physical interactions that are reversible and non-specific. Subsequently, specific, and irreversible interactions at the molecular and cellular level occurs where physical forces, such as gravitational forces, Brownian motion, Lifshitz–van der Waals attraction, hydrophobic interactions, and surface electrostatic charges, attract the bacteria towards the surface of the material. Following this attraction, adsorption of the cells, and attachment takes place. During the second stage, certain structures of bacteria, like the capsules, flagella, and nanofibers, establish a close and irreversible bond with the implant surface. Furthermore, the bare surface of the material quickly becomes coated with the protein of the extracellular matrix (ECM) adhesins; e.g., microbial surface components that recognize the adhesive matrix molecules, are crucial in mediating the binding between bacteria and the ECM proteins on the surface of the coating, promoting bacterial aggregation. In summary, bacterial adhesion is influenced by the characteristics of the bacteria, the properties of the material surface, conditions of the microenvironment, and hydrodynamic factors (32).
Formation of biofilms
Biofilm formation is a dynamic procedure that consists of several stages: adhesion of bacterial, formation of microcolony, maturation of biofilm, and finally dissipation of biofilm. Initially, individual, sparingly distributed bacteria gets adhered to the surface and progressively cluster for small colony formation. These colonies secrete an ECM that encases them. The biofilm then matures through signalling processes, developing a tower-like assembly that establishes a tight attachment to the substrate. This biofilm offers a protective 3-dimensional structure fostering the bacteria. Key factors contributing to the biofilm’s boosted antibiotic resistance and immunity to the host include biofilm network impermeability, horizontal transfer of genes, and the phenotypic variations between the different microbes within the substrate. ECM primarily consists of extracellular DNA (eDNA), proteins, extracellular polysaccharides, and teichoic acid. In the case of S. epidermidis and S. aureus, the polysaccharide intercellular adhesion expression and eDNA release are crucial mechanisms for the formation of biofilm. In unfavourable environments, a few bacterial cells may self-sacrifice to create a more appropriate living environment needed for other living bacteria. The autolyzed cells release the eDNA, which facilitates the maturation, and stabilizes the matrix of the biofilm (33–37). The mechanism of biofilm formation is shown in Figure 2 [adopted from (38)].
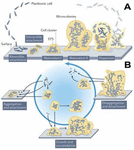
Figure 2. Stages of biofilm development: classic (A) and (B) modern [adapted from (38)].
Once the biofilm is formed, it becomes nearly impossible to treat the infection of the implant, leading to detrimental effects on the patient.
Interaction amid bacteria and the host cell
The pathogenic bacteria can access the prosthesis site in orthopedic IAI through both hematogenous (blood-borne) routes and contiguous spread. Cells of the innate immune system, such as macrophages and neutrophils, are employed at the site of infection via pattern recognition receptors, including Toll-like receptors, that can bind to the bacterial pathogen-associated molecular patterns. This binding activates the signalling of nuclear factor kappa B, leading to an inflammatory response (39). In this environment, immune cells secrete various cytokines and chemokines and employ mechanisms such as reactive oxygen species generation, phagocytosis, degranulation, antimicrobial peptides (AMPs), and neutrophil extracellular traps to engulf and eliminate the bacteria (40). Additionally, activation of the adaptive immune system can lead to antibody production, providing protection against recurrent infection for a long time. While effective clearance of planktonic bacteria is achieved by this type of immune response in the absence of implants, the presence of an implant triggers the innate immune response, as the implant itself gets recognized as a foreign body (41).
The above examples instigate the researchers to develop a functional coating on the implant material that can combat the evasion of bacteria, disrupt the formation of biofilms, and do not evoke an immune response against the host.
Functional coatings for orthopedic implants
There are many types of coating techniques. However, coatings applied under an electric field are used for the modification of the surface of the biomaterials. Recently, ceramic, metallic, polymer, and various composite electrodeposited coatings have been developed, each with distinct microstructures and properties (42–44).
Methods of synthesis
Methods such as direct cathodic electrodeposition, electrophoretic deposition, pulse cathodic deposition, plasma electrochemical oxidation in phosphate- and calcium-rich electrolytes, electro-discharge, and electro-spark techniques are employed to engineer these coatings. Of these, the most widely used are electrophoretic deposition and direct and pulse cathodic electrodeposition. Key factors like electrolyte composition, pH, potential and current, and temperature play a significant role in the coating process. Biocoatings, intended for biological applications, mostly employ metals, polymers, ceramics, bioglasses, or composites. They can be either co-deposited or surface-layered (hybrid or sandwich coatings) using various techniques, including the abovementioned methods and plasma vapor deposition, magnetron sputtering, chemical vapor deposition, and pulsed laser deposition. These coatings can be applied to solid or porous substrates (45). Various coatings and their synthesis methods are depicted in Figure 3.
Laser techniques-based surface treatments enhance the adhesion of coatings to substrates while improving the biological characteristics of functionalized medical devices without compromising their mechanical properties. Among these techniques, pulsed laser deposition, matrix-assisted laser deposition, and both simple and double laser writing stand out compared to additional well-known methods of deposition, such as 3D bioprinting, magnetron sputtering, extrusion, inkjet printing, dip coating, fused deposition modelling, and plasma spray. Each of these methods can be adapted for surface functionalization to modify the local morphology, crystal structure, and their chemistry, which in turn influences the behaviour of biomaterials for specific applications. Laser-based functionalization techniques of the surfaces can be precisely controlled in a confined area for effective delivery of the concentrated energy (46, 47).
Functional coating research
Orthopedic implants are often made from Ti, stainless steel, and CoCrMo alloys, but these materials can fail due to factors such as infection, corrosion, inflammation, stress shielding, elastic modulus mismatch, and wear, and tear. To improve the performance of implants, advancements in design, materials, and surface modifications have been developed, with coating techniques being particularly successful (48, 49). Techniques like physical vapor deposition, electric arc oxidation, chemical vapor deposition, sol-gel, and plasma spraying are used to enhance the biocompatibility, corrosion resistance, and improved mechanical properties of the metal implants. Coatings such as hydroxyapatite (HA), bioactive glass, and titanium nitride have shown substantial performance improvements (50, 51). Magnesium-based alloys coated with silk fibroin, Zr, and coatings with nanosilver and vitamin E on biodegradable implants have also been explored with improved properties. Nonetheless, issues related to the adhesion, stability, and degradation of these coatings still pose challenges for widespread industrial use. Recent research indicates that adding materials such as tantalum, chitosan, graphene oxide, biodegradable metals, and titanium dioxide to HA or biphasic calcium phosphate (BCP) coatings can enhance their properties (52–56). Moreover, hybrid coatings with inner layers have been shown to improve both biocompatibility and mechanical performance. Coating layers with releasable metal ions enhances bioactivity, with multiple ions synergistically boosting antibacterial properties and cellular compatibility. For instance, calcium (Ca) and magnesium (Mg) ions support osteoblast growth, while copper (Cu) and silver (Ag) ions offer strong antibacterial effects. Scientists have done extensive research on the advancement of functional antibacterial coatings on orthopedic implants (48, 57). Table 1 summarizes the recent developments in the field of functional antibacterial coatings. From the table, it is noticed that both organic and inorganic types of coatings were utilized for the developing antibacterial coatings over medical implants that could substantially avoid bacterial cell adhesion leading to IAIs. Other functional coatings are summarized in Table 2.
Potential clinical use of antimicrobial implants in orthopedics
Antimicrobial coatings of orthopedic implants are useful in reducing bacterial colonization and biofilm formation
on implant surfaces. Biofilms are difficult to eliminate, being structured communities of bacteria encased in a self-produced polymeric matrix, and account for the vast majority of chronic infections (34). By integrating antimicrobial agents into a coating, using, for instance, silver nanoparticles, quaternary ammonium compounds, or natural extracts that contain quercitrin, the adhesiveness of the bacteria can greatly be reduced, which in turn boosts the longevity as well as successful implementation of an orthopedic device (58). Silver coatings prevent the replication of bacteria, notably Staphylococcus aureus and Escherichia coli (50). Magnesium-based alloys will be able to provide temporary structural support and reduce infection risk. Magnesium alloys, as they biodegrade, ensure that the patients will have no more surgeries to remove permanent implants, which increases the outcome of the patients and decreases health care costs (59). In addition, the magnesium ions help in increasing the osteogenic activity, thus assisting in healing bone tissue (12). Despite this great potential of the antimicrobial implant, several challenges still remain. The demerit of these coatings is that the release of antibacterial agents may gradually decline with time, leading to infection (6). Further, some antimicrobial agents are also cytotoxic in nature. The regulatory approval for new coatings is a time-consuming and highly complex processes that requires in-vivo testing for the safety and efficacy of the product (20).
Comparative analysis of functional coatings on orthopedic implants
Table 3 presents a comparison of key performance metrics to give a clear picture of the effectiveness of different functional coatings. The table lists the antibacterial activity, biocompatibility, and economic viability of the coating materials taken into consideration, including chitosan-ZnO composites, magnesium-based alloys, and others.
Discussion
Antibacterial activity
Quercitrin-coated Ti-6Al-4V and the dual-functional coatings derived from quaternary ammonium salts present superior antibacterial activity; thereby, these coatings diminish the adhesion of bacteria as well as the formation of biofilms considerably. For instance, quercitrin, apart from improving the biocompatibility, induces osteogenic differentiation, and is considered to be an excellent potential for orthopedic use (60). However, the long-term stability of such coatings in the physiological environment is a concern, since the release of antibacterial agents may decrease with time, and recurrence of infection might be possible.
Biocompatibility
Vitamin E-coated Ti alloys exhibit excellent biocompatibility with anti-adhesive properties. They are thus essential for the use of temporary implants (61). On one hand, though they prevent the adherence of bacteria, they could impair the essential process of osseointegration in permanent implants for long-term successful integration with bones. There should be proper clinical assessment regarding these potential compromises where infection would be prevented with simultaneous promotion for integration with bones.
Mechanical properties
In this regard, hydroxyapatite coatings exhibit extraordinary bioactivity and have the ability to speed up the process of osseointegration. However, there are instances in which these coatings are brittle and are unable to withstand certain mechanical pressures that are placed in-vivo (50). On the other hand, polymers, particularly those composed of silk fibroin, are both flexible and durable. However, their long-term durability and wear resistance in a dynamic biological environment should be investigated further (59).
Scalability and cost
Coating technologies are also costly and difficult to implement on a large scale due to their high-cost constraints in clinical applications. Certainly, the high-efficiency techniques, such as magnetron sputtering and pulsed laser deposition, are remarkable (47). However, they are expensive and challenging to scale for mass production. Electrophoretic deposition and other older methods are cheaper and easier to use on a large scale, but they are not as good at evenly covering and sticking things together.
Challenges with regulatory approval
These coatings face clinical translation challenges with regulatory approval. As they often requires in-vivo testing in a multitude of animals and a complete checklist of approved protocols for safety and effectiveness to create new coatings possible for clinical usage, which can take its time (20).
Conclusion
The development of functional coatings on biometals is a viable technique for improving the performance and lifetime of orthopedic implants while also addressing the serious challenge faced by implant-related infections. This review focuses on the several strategic techniques that have been used to develop coatings that have antibacterial agents in them to avoid the formation of biofilms and the infections that could result from them, in addition to having mechanical attributes that are biocompatible. Advanced materials like biodegradable metals, ceramics, and polymers combined with cutting-edge coating processes have shown great promise in enhancing implantation and mitigating the negative side effects of commonly used metallic implants. Current studies on a variety of functional coatings, such as the recently developed bioactive ion release and biomedical natural chemicals, show that surface characteristics must be customized to satisfy the diverse requirements of orthopedic applications. As a result, hybrid coatings and their development of multipurpose materials pave the way for future advancements in implants that will benefit patients more, produce better results, and save healthcare costs. Addressing regulatory issues and ensuring the scalability of these coatings for clinical usage are crucial as the industry develops. Long-term in-vivo tests and the development of standardized procedures for evaluating the effectiveness and safety of these novel coatings should be the main objectives of future research.
References
1. Thanigaivel, S, Priya, AK, Balakrishnan, D, Dutta, K, Rajendran, S, and Soto-Moscoso, M. Insight on recent development in metallic biomaterials: strategies involving synthesis, types and surface modification for advanced therapeutic and biomedical applications. Biochem Eng J. (2022) 187:108522.
Rokaya, D, Bohara, S, Srimaneepong, V, Kongkiatkamon, S, Khurshid, Z, Heboyan, A, et al. Metallic biomaterials for medical and dental prosthetic applications. In: Jana S editor. Functional biomaterials: Drug delivery and biomedical applications. Singapore: Springer (2022). p. 503–22.
3. Sathishkumar, S, Paulraj, J, Chakraborti, P, and Muthuraj, M. Comprehensive review on biomaterials and their inherent behaviors for hip repair applications. ACS Appl Bio Mater. (2023) 6:4439–64.
4. Priyadarsini, S, Mukherjee, S, and Mishra, M. Nanoparticles used in dentistry: a review. J Oral Biol Craniofacial Res. (2018) 8:58–67.
5. Jiao, J, Zhang, S, Qu, X, and Yue, B. Recent advances in research on antibacterial metals and alloys as implant materials. Front Cell Infect Microbiol. (2021) 11:693939.
6. Feng, JB, Chen, R, Li, B, Jiang, BH, and Li, B. The current trend of antibacterial prostheses and prosthetic surface coating technologies to prevent prosthetic joint infection for artificial joint replacement. J Biomater Tissue Eng. (2023) 13:1046–60.
7. Wang, N, Ma, Y, Shi, H, Song, Y, Guo, S, and Yang, S. Mg-, Zn-, and Fe-based alloys with antibacterial properties as orthopedic implant materials. Front Bioeng Biotechnol. (2022) 10:888084.
8. Al-allaq, AA, Kashan, JS, Abdul-Kareem, FM, and Alani, AM. Review of in vivo investigations on metal implants for bone tissue engineering. Regen Eng Transl Med. (2024):1–25. doi: 10.1007/s40883-024-00352-2
9. Li, J, Qin, L, Yang, K, Ma, Z, Wang, Y, Cheng, L, et al. Materials evolution of bone plates for internal fixation of bone fractures: a review. J Mater Sci Technol. (2020) 36:190–208.
10. Paiva, JCC, Oliveira, L, Vaz, MF, and Costa-de-Oliveira, S. Biodegradable bone implants as a new hope to reduce device-associated infections—a systematic review. Bioengineering. (2022) 9:409.
11. Rahim, SA, Joseph, MA, and Sampath Kumar, TS. Recent progress in surface modification of Mg alloys for biodegradable orthopedic applications. Front Mater. (2022) 9:848980.
12. Mishra, TN, Kumar, A, and Prasad, SB. Magnesium based implants: alloying and coating strategies for improvement in its biomechanical and biocorrosion properties. Proc Inst Mech Eng Part L J Mater Des Appl. (2024):14644207241269608. doi: 10.1177/146442072412696
13. Tsakiris, V, Tardei, C, and Clicinschi, FM. Biodegradable Mg alloys for orthopedic implants–a review. J Magnes Alloy. (2021) 9:1884–905.
14. Rouf, S, Malik, A, Raina, A, Haq, MIU, Naveed, N, Zolfagharian, A, et al. Functionally graded additive manufacturing for orthopedic applications. J Orthop. (2022) 33:70–80.
15. Shi, H, Zhou, P, Li, J, Liu, C, and Wang, L. Functional gradient metallic biomaterials: techniques, current scenery, and future prospects in the biomedical field. Front Bioeng Biotechnol. (2021) 8:616845.
Keerthiga, G, Simi, VS, and Mohan, L. Surface modifications of biometals. In: Kaya S, Yesudass S, Arthanari S, Bose S, and Serdaroğlu G editors. Materials development and processing for biomedical applications. Boca Raton, FL: CRC Press (2022). p. 197–210.
Lakhani, A, and Khatri, K. Orthopedic applications of implant coatings. In: ul Islam S, Hussain CM, and Shukla SK editors. Antiviral and antimicrobial coatings based on functionalized nanomaterials. Amsterdam: Elsevier (2023). p. 589–604.
18. Yang, X, Xiong, S, Zhou, J, Zhang, Y, He, H, Chen, P, et al. Coating of manganese functional polyetheretherketone implants for osseous interface integration. Front Bioeng Biotechnol. (2023) 11:1182187.
19. Kim, S, Chen, JB, and Clifford, A. Tuning the biointerface: low-temperature surface modification strategies for orthopedic implants to enhance osteogenic and antimicrobial activity. ACS Appl Bio Mater. (2021) 4:6619–29.
20. Syeddan, SA. Research methodology and mechanisms of action of current orthopaedic implant coatings. J Long Term Eff Med Implants. (2023) 33:51–66.
23. Claes, L, Recknagel, S, and Ignatius, A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. (2012) 8:133–43.
24. Einhorn, TA, and Gerstenfeld, LC. Fracture healing: mechanisms and interventions. Nat Rev Rheumatol. (2015) 11:45–54.
25. Ono, T, and Takayanagi, H. Osteoimmunology in bone fracture healing. Curr Osteoporos Rep. (2017) 15:367–75.
26. Kolar, P, Gaber, T, Perka, C, Duda, GN, and Buttgereit, F. Human early fracture hematoma is characterized by inflammation and hypoxia. Clin Orthop Relat Res. (2011) 469:3118–26.
27. Girigoswami, K, Devender, NS, and Girigoswami, A. Fate of stem cells grown on the extracellular matrix isolated from cancer cells and their possible applications in tissue engineering. Curr Sci. (2021) 120:1616.
28. Girigoswami, K, Saini, D, and Girigoswami, A. Extracellular matrix remodeling and development of cancer. Stem Cell Rev Rep. (2021) 17:739–47.
29. Papachristou, DJ, Georgopoulos, S, Giannoudis, PV, and Panagiotopoulos, E. Insights into the cellular and molecular mechanisms that govern the fracture-healing process: a narrative review. J Clin Med. (2021) 10:3554.
30. Li, H, Yao, Z, Zhang, J, Cai, X, Li, L, Liu, G, et al. The progress on physicochemical properties and biocompatibility of tantalum-based metal bone implants. SN Appl Sci. (2020) 2:1–14.
31. Jacobs, A, Renaudin, G, Forestier, C, Nedelec, JM, and Descamps, S. Biological properties of copper-doped biomaterials for orthopedic applications: a review of antibacterial, angiogenic and osteogenic aspects. Acta Biomater. (2020) 117:21–39.
32. Filipović, U, Dahmane, RG, Ghannouchi, S, Zore, A, and Bohinc, K. Bacterial adhesion on orthopedic implants. Adv Colloid Interface Sci. (2020) 283:102228.
33. Zhang, K, Li, X, Yu, C, and Wang, Y. Promising therapeutic strategies against microbial biofilm challenges. Front Cell Infect Microbiol. (2020) 10: 359.
34. Arciola, CR, Campoccia, D, and Montanaro, L. Implant infections: adhesion, biofilm formation and immune evasion. Nat Rev Microbiol. (2018) 16:397–409.
35. Makabenta, JM, Nabawy, A, Li, CH, Schmidt-Malan, S, Patel, R, and Rotello, VM. Nanomaterial-based therapeutics for antibiotic-resistant bacterial infections. Nat Rev Microbiol. (2021) 19:23–36.
36. Flemming, HC, Wingender, J, Szewzyk, U, Steinberg, P, Rice, SA, and Kjelleberg, S. Biofilms: an emergent form of bacterial life. Nat Rev Microbiol. (2016) 14:563–75.
37. Okshevsky, M, and Meyer, RL. The role of extracellular DNA in the establishment, maintenance and perpetuation of bacterial biofilms. Crit Rev Microbiol. (2015) 41:341–52.
38. Perchikov, R, Cheliukanov, M, Plekhanova, Y, Tarasov, S, Kharkova, A, Butusov, D, et al. Microbial biofilms: features of formation and potential for use in bioelectrochemical devices. Biosensors. (2024) 14: 302.
39. Fournier, B. The function of TLR2 during staphylococcal diseases. Front Cell Infect Microbiol. (2013) 2:167.
40. Amin Yavari, S, Castenmiller, SM, van Strijp, JA, and Croes, M. Combating implant infections: shifting focus from bacteria to host. Adv Mater. (2020) 32:2002962.
41. Seebach, E, and Kubatzky, KF. Chronic implant-related bone infections—can immune modulation be a therapeutic strategy? Front Immunol. (2019) 10:1724.
42. Chowdhury, S, and Arunachalam, N. Surface functionalization of additively manufactured titanium alloy for orthopaedic implant applications. J Manuf Process. (2023) 102:387–405.
43. Al-Shalawi, FD, Mohamed Ariff, AH, Jung, DW, Mohd Ariffin, MKA, Seng Kim, CL, Brabazon, D, et al. Biomaterials as implants in the orthopedic field for regenerative medicine: metal versus synthetic polymers. Polymers (Basel). (2023) 15:2601.
44. Grigoriev, S, Sotova, C, Vereschaka, A, Uglov, V, and Cherenda, N. Modifying coatings for medical implants made of titanium alloys. Metals (Basel). (2023) 13:718.
45. Zielinski, A, and Bartmanski, M. Electrodeposited biocoatings, their properties and fabrication technologies: a review. Coatings. (2020) 10:782.
46. Badiceanu, M, Anghel, S, Mihailescu, N, Visan, AI, Mihailescu, CN, and Mihailescu, IN. Coatings functionalization via laser versus other deposition techniques for medical applications: a comparative review. Coatings. (2022) 12:71.
47. Meng, M, Wang, J, Huang, H, Liu, X, Zhang, J, and Li, Z. 3D printing metal implants in orthopedic surgery: methods, applications and future prospects. J Orthop Transl. (2023) 42:94–112.
48. Hussain, M, Askari Rizvi, SH, Abbas, N, Sajjad, U, Shad, MR, Badshah, MA, et al. Recent developments in coatings for orthopedic metallic implants. Coatings. (2021) 11:791.
49. Solangi, NH, Karri, RR, Mubarak, NM, and Mazari, SA. Mechanism of polymer composite-based nanomaterial for biomedical applications. Adv Ind Eng Polym Res. (2024) 7:1–19.
50. Nawaz, MH, Aizaz, A, Ropari, AQ, Shafique, H, Imran, OB, Minhas, BZ, et al. A study on the effect of bioactive glass and hydroxyapatite-loaded Xanthan dialdehyde-based composite coatings for potential orthopedic applications. Sci Rep. (2023) 13:17842.
51. Palierse, E, Roquart, M, Norvez, S, and Corté, L. Coatings of hydroxyapatite–bioactive glass microparticles for adhesion to biological tissues. RSC Adv. (2022) 12:21079–91.
52. Tan, J, Li, L, Li, B, Tian, X, Song, P, and Wang, X. Titanium surfaces modified with graphene oxide/gelatin composite coatings for enhanced antibacterial properties and biological activities. ACS Omega. (2022) 7:27359–68.
53. Silva, AO, Cunha, RS, Hotza, D, and Machado, RAF. Chitosan as a matrix of nanocomposites: a review on nanostructures, processes, properties, and applications. Carbohydr Polym. (2021) 272:118472.
54. Bouammali, H, Bourassi, L, Bouammali, B, Siaj, M, Challioui, A, and Touzani, R. Graphene and chitosan innovative materials for water treatment. Mater Today Proc. (2023) 72:3577–88.
55. Nie, L, Hou, M, Wang, T, Sun, M, and Hou, R. Nanostructured selenium-doped biphasic calcium phosphate with in situ incorporation of silver for antibacterial applications. Sci Rep. (2020) 10:13738.
56. Ebrahimi, M, Botelho, MG, and Dorozhkin, SV. Biphasic calcium phosphates bioceramics (HA/TCP): concept, physicochemical properties and the impact of standardization of study protocols in biomaterials research. Mater Sci Eng C. (2017) 71:1293–312.
57. Tripathi, S, Raheem, A, Dash, M, Kumar, P, Elsebahy, A, Singh, H, et al. Surface engineering of orthopedic implants for better clinical adoption. J Mater Chem B. (2024) 12:11302–35.
58. Ghilini, F, Fagali, N, Pissinis, DE, Benítez, G, and Schilardi, PL. Multifunctional titanium surfaces for orthopedic implants: antimicrobial activity and enhanced osseointegration. ACS Appl Bio Mater. (2021) 4:6451–61.
59. Li, H, Si, S, Yang, K, Mao, Z, Sun, Y, Cao, X, et al. Hexafluoroisopropanol based silk fibroin coatings on AZ31 biometals with enhanced adhesion, corrosion resistance and biocompatibility. Prog Org Coatings. (2023) 184:107881.
60. Llopis-Grimalt, MA, Arbós, A, Gil-Mir, M, Mosur, A, Kulkarni, P, Salito, A, et al. Multifunctional properties of quercitrin-coated porous Ti-6Al-4V implants for orthopaedic applications assessed in vitro. J Clin Med. (2020) 9:855.
61. Gamna, F, Cochis, A, Scalia, AC, Vitale, A, Ferraris, S, Rimondini, L, et al. The use of vitamin E as an anti-adhesive coating for cells and bacteria for temporary bone implants. Surf Coatings Technol. (2022) 444: 128694.
62. Parfenova, LV, Galimshina, ZR, Gil’fanova, GU, Alibaeva, EI, Danilko, KV, Pashkova, TM, et al. Hyaluronic acid bisphosphonates as antifouling antimicrobial coatings for PEO-modified titanium implants. Surf Interfaces. (2022) 28:101678.
63. Chen, C, Li, Z, Li, X, Kuang, C, Liu, X, Song, Z, et al. Dual-functional antimicrobial coating based on the combination of zwitterionic and quaternary ammonium cation from rosin acid. Compos Part B Eng. (2022) 232:109623.
64. Zermeño-Pérez, D, Chouirfa, H, Rodriguez, BJ, Dürig, T, Duffy, P, and Cróinín, TÓ. Bioresorbable polyester coatings with antifouling and antimicrobial properties for prevention of biofilm formation in early stage infections on Ti6Al4V hard-tissue implants. ACS Appl Bio Mater. (2024) 7:5728–39.
65. Acosta, S, Ibañez-Fonseca, A, Aparicio, C, and Rodríguez-Cabello, JC. Antibiofilm coatings based on protein-engineered polymers and antimicrobial peptides for preventing implant-associated infections. Biomater Sci. (2020) 8:2866–77.
66. Gouveia, Z, Perinpanayagam, H, and Zhu, J. Development of robust chitosan–silica class II hybrid coatings with antimicrobial properties for titanium implants. Coatings. (2020) 10:534.
67. Ye, Z, Sang, T, Li, K, Fischer, NG, Mutreja, I, Echeverría, C, et al. Aparicio, Hybrid nanocoatings of self-assembled organic-inorganic amphiphiles for prevention of implant infections. Acta Biomater. (2022) 140:338–49.
68. Lin, MH, Wang, YH, Kuo, CH, Ou, SF, Huang, PZ, Song, TY, et al. Hybrid ZnO/chitosan antimicrobial coatings with enhanced mechanical and bioactive properties for titanium implants. Carbohydr Polym. (2021) 257:117639.
69. Aslan, AK, Bahce, E, and Güler, MS. Electrochemical corrosion and metal ion release protective efficiency of the multilayer TaN coatings on CoCrMo biomedical alloy. Mater Sci. (2021) 27:416–24.
70. Camargo, ER, Serafim, BM, da Cruz, AF, Soares, P, de Oliveira, CC, Saul, CK, et al. Bioactive response of PMMA coating obtained by electrospinning on ISO5832-9 and Ti6Al4V biomaterials. Surf Coatings Technol. (2021) 412:127033.
71. Fayed, O, van Griensven, M, Tahmasebi Birgani, Z, Plank, C, and Balmayor, ER. Transcript-activated coatings on titanium mediate cellular osteogenesis for enhanced osteointegration. Mol Pharm. (2021) 18: 1121–37.
72. Zhu, X, Shi, Y, Yang, L, Chen, Q, Luo, X, Zhang, Q, et al. Creation of bioactive ceramic composite coatings on Zn–Mn–Mg alloy via micro-arc oxidation and hydrothermal treatment for orthopedic implant applications. ACS Appl Eng Mater. (2023) 1:734–43.
73. Qian, J, Qin, H, Zeng, P, Hou, J, Mo, X, Shen, G, et al. Metal-organic Zn-zoledronic acid and 1-hydroxyethylidene-1, 1-diphosphonic acid nanostick-mediated zinc phosphate hybrid coating on biodegradable Zn for osteoporotic fracture healing implants. Acta Biomater. (2023) 166:685–704.
74. Benzarti, Z, Itani, S, Castro, JD, Carvalho, S, and Ramos, AS. Design multifunctional Mg–Zr coatings regulating Mg alloy bioabsorption. J Magnes Alloy. (2024) 12:1461–78.
75. Li, H, Ma, T, Zhang, J, Mao, Z, Liang, H, Sun, Y, et al. Boosting corrosion resistance and osteogenic activity of magnesium-based implants with metal ions chelated silk fibroin coating via binary solvent system. Chem Eng J. (2024) 485:149905.
© The Author(s). 2024 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.